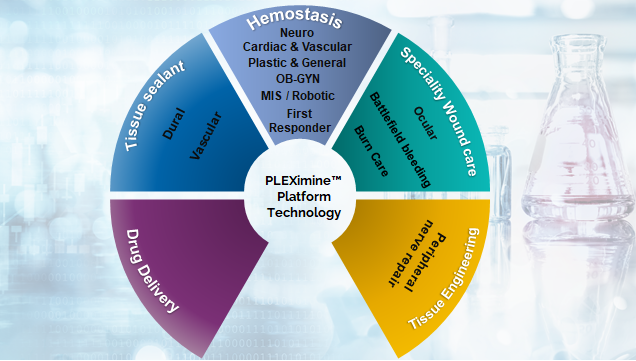
Hemostasis
The first spin off from the PLEXimine™ platform is PlexiClot™ Absorbable Hemostat, a groundbreaking device targeted initially at brain and spine surgery. PlexiClot is a handheld device that delivers processed polysaccharide solutions as a spray onto a bleeding site in the brain or spine. The spray forms a 3-D structured and dynamic hydrogel device in situ in 2-9 seconds. The patented chemistry forms a physical device with the first fundamental innovations in mechanisms of action for absorbable hemostats since the current standard of care was introduced in 1945. These new mechanisms of action provide IP-protected strategic advantages in the $1.9B market for absorbable hemostats in neurosurgery.
- PlexiClot provides a tamponade that stops bleeding in about 20 seconds. It’s patented 3-D structure produces coagulation with a stable clot shortly afterward. This speed of action is 15x-30x faster than the standard care. Since absorbable hemostats are often applied several times per case, PlexiClot can save 20-30 minutes per case compared to current techniques. PlexiClot can reduce OR costs and the complications and infections associated with longer cases.
- All current absorbable hemostats swell in use , 20% – 200%. In neurosurgery, this device swelling can cause mass effects that produce pain and paralysis that require re-operation to alleviate. This problem with absorbable hemostats has been documented in dozens of clinical publications and in the FDA MAUDE database. This swelling does not lead to problems routinely, but it is a routine threat that surgeons must address in every case. PlexiClot eliminates this threat by gently shrinking, a patented feature, instead of swelling.
- Pressure or manual compression on brain or spinal nerve tissue can produce post-op neurological deficits. All current absorbable hemostats are applied with some degree of manual pressure in order to be effective. PlexiClot performs without any manual pressure or compression, eliminating this threat with patented technology.
- All current devices are opaque, and surgeons must often be forced to judge indirectly if hemostasis has been achieved. PlexiClot is transparent and allows surgeons to verify that hemostasis has been achieved in real time.
After neurosurgery, PlexiClot will be launched into other surgical subspecialties, such as gynecology and cardiac procedures, plastic, orthopaedic and general surgery. PlexiClot can be delivered as a flowable liquid for laparoscopic, endoscopic and robotic applications.
Endomedix is currently exploring applications for PlexiClot in military and first responder applications.
Other PLEXimine Applications
There are several other applications of PLEXimine technology, beyond hemostasis intended uses. These include:
- Tissue sealants – especially for dura mater applications in neurosurgery, vascular sealant applications and ocular applications
- Tissue engineering devices – to deliver emerging cellular therapies
- Intraoperative drug delivery
- Specialty wound care – burn care, ocular applications
